Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 and Eu(As
and Eu(As P
P )
)
Onuki, Yoshichika*; Karube, Kosuke*; Aoki, Dai*; Nakamura, Ai*; Homma, Yoshiya*; Matsuda, Tatsuma*; Haga, Yoshinori; Takeuchi, Tetsuya*
Journal of the Physical Society of Japan, 92(11), p.114703_1 - 114703_12, 2023/11
Times Cited Count:0 Percentile:0(Physics, Multidisciplinary)Villaret, F.*; Boulnat, X.*; Aubry, P.*; Yano, Yasuhide; Otsuka, Satoshi; Fabregue, D.*; de Carlan, Y.*
Materials Science & Engineering A, 824, p.141794_1 - 141794_10, 2021/09
Times Cited Count:3 Percentile:24.75(Nanoscience & Nanotechnology)Kakihana, Masashi*; Nishimura, Kengo*; Takeuchi, Tetsuya*; Haga, Yoshinori; Harima, Hisatomo*; Hedo, Masato*; Nakama, Takao*; Onuki, Yoshichika*
Journal of the Physical Society of Japan, 88(4), p.044712_1 - 044712_11, 2019/04
Times Cited Count:4 Percentile:36.25(Physics, Multidisciplinary) -LiAl; Relation to the defect structure
-LiAl; Relation to the defect structureSugai, Hiroyuki
Solid State Ionics, 177(39-40), p.3507 - 3512, 2007/01
The diffusion coefficient and its activation energy (116.3  11.7 kJ/mol) of tritium in an intermetallic compound
11.7 kJ/mol) of tritium in an intermetallic compound  -LiAl are determined at temperatures from 700 to 848 K. Though the present result for the diffusion coefficient is almost the same as that reported previously, the present result for the activation energy turns out nearly twice of that (64.9
-LiAl are determined at temperatures from 700 to 848 K. Though the present result for the diffusion coefficient is almost the same as that reported previously, the present result for the activation energy turns out nearly twice of that (64.9  3.8 kJ/mol). The present result for the activation energy is consistent with the systematics that an increase of lithium concentration in Al-Li systems increases the activation energy, but the previous result is not. Furthermore, a consideration of the crystal structure and defect structure suggests that tritium diffuses and is impeded by the attractive interaction with lithium atom at lithium sublattices.
3.8 kJ/mol). The present result for the activation energy is consistent with the systematics that an increase of lithium concentration in Al-Li systems increases the activation energy, but the previous result is not. Furthermore, a consideration of the crystal structure and defect structure suggests that tritium diffuses and is impeded by the attractive interaction with lithium atom at lithium sublattices.
 -LiAl; Relation to the defect structure
-LiAl; Relation to the defect structureSugai, Hiroyuki
Solid State Ionics, 177(39-40), p.3507 - 3512, 2007/01
Times Cited Count:3 Percentile:17.83(Chemistry, Physical)The diffusion coefficients and its activation energy (103.7 9.5 kJ/mol) for tritium in intermetallic compound
9.5 kJ/mol) for tritium in intermetallic compound  -LiAl are determined at temperatures from 699 to 886 K. Though the present result for the diffusion coefficient is almost the same as that reported earlier, the activation energy turns out nearly twice of that (64.9
-LiAl are determined at temperatures from 699 to 886 K. Though the present result for the diffusion coefficient is almost the same as that reported earlier, the activation energy turns out nearly twice of that (64.9 3.8 kJ/mol) reported earlier. On the basis of the crystal structure and defect structure, the large activation energy of this study suggest that tritium diffuses interstitially and is impeded by an attractive interaction with lithium atoms in lithium sublattices.
3.8 kJ/mol) reported earlier. On the basis of the crystal structure and defect structure, the large activation energy of this study suggest that tritium diffuses interstitially and is impeded by an attractive interaction with lithium atoms in lithium sublattices.
 Pb
Pb intermetallic compound
intermetallic compoundOno, Masao; Huang, X. S.*; Shibata, Yasuhiro*; Iguchi, Yusuke*; Sakai, Seiji; Maekawa, Masaki; Chen, Z. Q.*; Osakabe, Toyotaka; Kawasuso, Atsuo; Naramoto, Hiroshi*; et al.
Proceedings of 1st International Conference on Diffusion in Solids and Liquids (DSL 2005), p.531 - 533, 2005/07
Recently, we formed atomic-scale graded structures in some miscible alloys and observed the decomposition in Bi Pb
Pb intermetallic compound by sedimentation of atoms under strong gravitational field. In this study, we measured positron lifetime of centrifuged Bi
intermetallic compound by sedimentation of atoms under strong gravitational field. In this study, we measured positron lifetime of centrifuged Bi Pb
Pb , to which the composition change was very small as it was treated at low temperature. It was found that the positron lifetime became longer than that of starting state. This indicated that the point defects (vacancy or divacancy) increased in the sample by centrifugal treatment. We are now investigating the relationship between increase in point defects and sedimentation of atoms.
, to which the composition change was very small as it was treated at low temperature. It was found that the positron lifetime became longer than that of starting state. This indicated that the point defects (vacancy or divacancy) increased in the sample by centrifugal treatment. We are now investigating the relationship between increase in point defects and sedimentation of atoms.
Yutani, Toshiaki*; Nakamura, Hiroo; Sugimoto, Masayoshi
JAERI-Tech 2005-036, 10 Pages, 2005/06
In the high flux region of the International Fusion Materials Irradiation Facility (IFMIF), the neutron irradiation damage for iron-based alloys will exceed 20 dpa/ year. An accurate specimen temperature measurement under a large amount of nuclear heating is a key issue but the change of heat transfer of gap between irradiation specimens and specimen holder during irradiation test is inevitable, if gap is filled with an inert gas and temperature is monitored by a thermocouple buried in the specimen holder. A solution to make heat transfer predictable is to fill the gap with a liquid metal (sodium or sodium-potassium alloy). An issue of compatibility between Reduced Activation Ferritic/Martensitic steels and the liquid metalsis addressed in this paper, and some recommendations for designing irradiation rig are presented, such as a purification control before filling liquid metals, or a careful selection of material of rig to avoid carbon mass transfer.
 C and 550
C and 550 C
CKurata, Yuji; Futakawa, Masatoshi; Saito, Shigeru
JAERI-Research 2005-002, 37 Pages, 2005/02
Static corrosion tests of various austenitic and ferritic/martensitic steels were conducted in oxygen-saturated liquid Pb-Bi at 450 C and 550
C and 550 C for 3000h to study the effects of temperature and alloying elements on corrosion behavior. Oxidation, grain boundary corrosion, dissolution and penetration were observed. The corrosion depth decreases at 450
C for 3000h to study the effects of temperature and alloying elements on corrosion behavior. Oxidation, grain boundary corrosion, dissolution and penetration were observed. The corrosion depth decreases at 450 C with increasing Cr content in steels regardless of ferritic/martensitic or austenitic steels. Appreciable dissolution of Ni and Cr does not occur in the three austenitic steels at 450
C with increasing Cr content in steels regardless of ferritic/martensitic or austenitic steels. Appreciable dissolution of Ni and Cr does not occur in the three austenitic steels at 450 C. The corrosion depth of ferritic/martensitic steels also decreases at 550
C. The corrosion depth of ferritic/martensitic steels also decreases at 550 C with increasing Cr content whereas the corrosion depth of austenitic steels, JPCA and 316ss becomes larger due to ferritization caused by dissolution of Ni at 550
C with increasing Cr content whereas the corrosion depth of austenitic steels, JPCA and 316ss becomes larger due to ferritization caused by dissolution of Ni at 550 C than that of ferritic/martensitic steels. An austenitic stainless steel containing about 5%Si exhibits fine corrosion resistance at 550
C than that of ferritic/martensitic steels. An austenitic stainless steel containing about 5%Si exhibits fine corrosion resistance at 550 C because the protective Si oxide film is formed and prevents dissolution of Ni and Cr.
C because the protective Si oxide film is formed and prevents dissolution of Ni and Cr.
Haruyama, Mitsuo; Takase, Misao*; Tobita, Hiroshi; Mori, Takamasa
Nihon Genshiryoku Gakkai Wabun Rombunshi, 3(2), p.185 - 192, 2004/06
no abstracts in English
The Working Team for Examination of the Sample from Core Shrouds and Primary Loop Recirculation Pipi; Nakajima, Hajime*; Shibata, Katsuyuki; Tsukada, Takashi; Suzuki, Masahide; Kiuchi, Kiyoshi; Kaji, Yoshiyuki; Kikuchi, Masahiko; Ueno, Fumiyoshi; Nakano, Junichi; et al.
JAERI-Tech 2004-015, 114 Pages, 2004/03
The Tokyo Electric Power Company (TEPCO) visually inspected the weld joint of core shroud at Fukushima Dai-ni Nuclear Power Station Unit-2 by a direction of the Nuclear and Industrial Agency, cracks were observed at outer side of the ring weld joint (H3) between a core shroud middle trunk and a middle ring. TEPCO has conducted a material examination with Nippon Nuclear Fuel Development Co. Ltd. (NFD) on the specimen including cracks sampled from the core shroud. The present examination has been performed with the objective to independently investigate and evaluate the materials by jointly attending the examination with NFD from the planning stage. Based on results of the present examination, the probable presence of tensile residual stress by welding process and dissolved oxygen contents in the cooling water, it was shown that the cracks were considered to be stress corrosion cracking (SCC). However, the cause of the cracks needs more consideration on the way of shroud construction.
Takeda, Tetsuaki
Proceedings of 12th International Conference on Nuclear Engineering (ICONE-12) (CD-ROM), 4 Pages, 2004/00
no abstracts in English
 FeCoO
FeCoO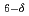 and SrBaFeCoO
and SrBaFeCoO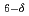 ; Effects induced by magnetic order and transport coherence
; Effects induced by magnetic order and transport coherenceRykov, A. I.*; Nomura, Kiyoshi*; Sawada, Tsuguo*; Mitsui, Takaya; Seto, Makoto*; Tamegai, Tsuyoshi*; Tokunaga, Masashi*
Physical Review B, 68(22), p.224401_1 - 224401_7, 2003/12
Times Cited Count:8 Percentile:42.85(Materials Science, Multidisciplinary)no abstracts in English
Kato, Chiaki
JAERI-Research 2003-013, 143 Pages, 2003/08
This study is investigation about stress corrosion cracking (SCC) of zirconium in nuclear fuel reprocessing. Chapter 1 is described background. Chapter 2 is explained experimental apparates. Chapter 3 is described the increased oxidization potential on the heat-transfer surface and suggested the initiation of SCC on a boiling heat-transfer surface. Chapter 4 is described that the SCC susceptibility increased with increasing nitric acid concentration and solution temperature on notched specimen by SSRT. In addition, the SCC susceptibility effected by the crystal anisotropy by the hot rolling direction and increased on a parallel face to the rolling direction. Chapter 5 is described that the SCC susceptibility increased in HAZ/base metal boundary in order to the preferential orientation of cleavage plane (0002). Chapter 6 is described that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition on the surface and the removal of the decomposition product from solution by boiling bubbles.
Okoshi, Minoru
Dekomisshoningu Giho, (26), p.2 - 12, 2002/11
The concept of clearance has been introduced by the International Atomic Energy Agency in 1996 and is very useful for the management of very low-level solid materials generating from the decommissioning of nuclear facilities. Therefore, the European Commission (EC) derived the specific clearance levels for metals, buildings and building rubble in RP 89 and 113, respectively. The EC also derived the general clearance levels for all solid materials generating from the regulated facilities in RP 122. Comparing the clearance levels of Japan with the unrounded levels of EC, the differences of levels are small. The biggest difference is found in Fe-55 and the EC's clearance level is about one fifth of Japanese clearance level. This is caused because the EC considers the direct ingestion of cleared building rubble by children and used the conservative ingestion rate of it. EC's discussions related to the clearance levels are very useful for Japan to derive our own clearance levels and to clear materials from regulatory control.
Mishima, Kaichiro*; Saito, Yasushi*
JAERI-Tech 2002-014, 83 Pages, 2002/03
no abstracts in English
 Cs following the decay of
Cs following the decay of  Xe atoms implanted in metals
Xe atoms implanted in metals*; *; *; *; *; *; Muto, Suguru*; Koizumi, Mitsuo; Osa, Akihiko; Sekine, Toshiaki; et al.
Journal of Radioanalytical and Nuclear Chemistry, 239(2), p.251 - 255, 1999/00
Times Cited Count:0 Percentile:0.01(Chemistry, Analytical)no abstracts in English
Nakayama, Shinichi; Yamaguchi, Tetsuji; Sekine, Keiichi
Radiochimica Acta, 74(1), p.15 - 19, 1996/01
no abstracts in English
J.K.Gibson*; R.G.Haire*; E.C.Beahm*; M.M.Gensini*; Maeda, Atsushi; Ogawa, Toru
Journal of Nuclear Materials, 211, p.215 - 222, 1994/00
Times Cited Count:9 Percentile:63.31(Materials Science, Multidisciplinary)no abstracts in English
Chihara, Junzo; *
Molecular Simulation, 12(3-6), p.187 - 195, 1994/00
Times Cited Count:3 Percentile:14.83(Chemistry, Physical)no abstracts in English
Nakamura, Hisashi; Kanazawa, Katsuo; Sato, Takayuki; ; Fujiki, Kazuo
Dekomisshoningu Giho, (9), p.41 - 50, 1993/12
no abstracts in English